As an organism develops, cells often migrate from one part of the body to another in order to end up in the right place within the body plan. However, the extent to which cells can ensure the accuracy of their final positions, and how external perturbations affect this accuracy, are poorly understood.
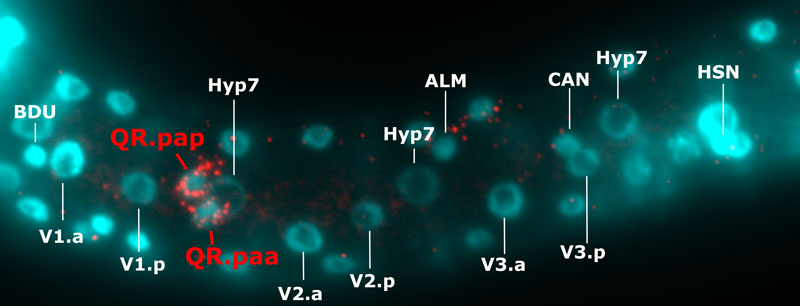
Figure: The migration of QR.pa stops upon its expression of the mig-1 gene, which is induced by a timing mechanism. The picture depicts mig-1 expression in the QR.pa daughters, in a larva of C. elegans wild isolate XZ1516. Single mig-1 mRNAs are displayed in red (single-molecule in situ hybridization) and cell nuclei in blue (DAPI). Positions of V seam cell and hyp7 nuclei were used to quantify the final position of QR.paa, QR.pap and other migrating neurons (BDU, ALM, CAN and HSN). Picture: Clément Dubois.
In the Caenorhabditis elegans roundworm, a particular cell that will become a neuron migrates from the tail to the head of the worm during the larval stage of development. Previous work from our HFSP co-investigator Rik Korswagen had shown that this cell stops migrating based on its own internal timer, not based on spatial cues. Therefore, we hypothesized that if the worm body were smaller, the cell would migrate further, and vice versa. Indeed, this is what we saw when we changed the body size, both by using genetic mutations and by using mother worms of different ages (which is known to affect body size).
However, the change in the final cell position was not as pronounced as one would expect if the cell were migrating at a fixed speed for a fixed amount of time. Therefore, we measured the migration speed directly and indeed found partial compensation for body size: in larger worms, the speed was somewhat higher. By incorporating the speed dependence into a simple mathematical model that also accounted for larval growth, we were able to explain the data with no free parameters.
Our results show that spatial perturbations at the organismal level can change spatial arrangement at the cell level, especially when cell position is based on a temporal, not spatial, mechanism. Nevertheless, our results also show that cells can partially correct for this change. The correction mechanism may be due to correlations between body size, cell size, and cell speed, or to a partial reliance on spatial cues, but in general it is unknown. This is an exciting direction for future research.
HFSP funding was critical in developing our collaboration and getting this project off the ground. The PIs would not have met and begun working together if not motivated by the unique opportunity presented by HFSP.


































