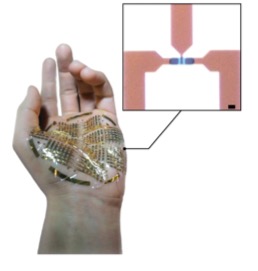
Left: Optical micrograph displaying the top view of an individual e-IGT (top). Ultra-flexible, ultra-thin e- IGT array conforming to the surface of a human hand (bottom). Scale bar, 5μm.
The challenge
Bioelectronic devices have numerous potential benefits for human health, from in-home wellness monitoring to diagnosis and treatment of neuropsychiatric diseases. However, safe and effective use of these devices is limited by the rigid, non-biocompatible electronic components that have to be incorporated to allow execution of the required functions. Specifically, these devices must be fast and sensitive to interact with the rapid, low amplitude signals generated by neural tissue. They should also be biocompatible, soft, and exhibit long-term stability in physiologic environments.
The approach
We developed a soft ion driven transistor (enhancement mode internal ion-gated electrochemical transistor (e-IGT)) that enables long-term stable and high performance operation in physiological conditions without the need of bulky and rigid encapsulation. Conventionally, transistors are made out of silicon (Si) but they require strong encapsulation in physiologic environments because ion diffusion damages the silicon components. These properties also restrict the abiotic/biotic coupling to capacitive interaction between the surface of the gate material and electrolyte. Organic materials have been used to overcome these limitations as they are inherently flexible but the electrical performance of these devices was insufficient for performing real-time brain signal recording and processing. In our devices, we take advantage of both electronic and ionic conduction of organic material to create transistors that have embedded mobile ions inside their channel. In this way, they are able to be switched in a fast and efficient manner.
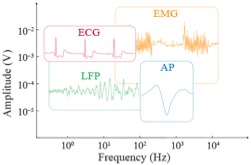
Above: Sample traces of in vivo signals acquired by e-IGTs plotted to reflect the wide span of frequency and amplitude characteristics.
The results
IGT time responses depend on hole (electronic) rather than ion mobility even though ions are slower. This is because in our transistors, ions are inside the channel and they do not need to travel long distances to participate in the channel switching process. We used these transistors to acquire a wide range of electrophysiological signals, including in vivo recording of neural action potentials, and to create soft, biocompatible, long-term implantable neural processing units for real-time detection of epileptic discharges. E-IGTs offer a safe, reliable, and high-performance building block for chronically implanted bioelectronics, with spatiotemporal resolution to the scale of individual neurons.
Applications and impact
E-IGTs are composed of only biocompatible and commercially available materials that do not require encapsulation in biological environments and are not impaired by exposure to water or ions. The composite material of the channel can be reproducibly manufactured in large quantities and is solution processible, making it more accessible to a broad range of fabrication processes. They are flexible and compatible with integration into a wide variety of conformable plastic substrates and have low leakage current, permitting fabrication of efficient integrated circuits. Sensing applications that require sparse detection of analyte over long periods of time would benefit from application of e-IGT because a transistor that is normally OFF does not consume extensive power. Overall, we have shown that e-IGTs can serve as reliable components for bioelectronic devices, with the potential to improve our ability to transduce and modulate physiologic signals. E-IGTs are also suitable for translation to use in humans, offering a safe, reliable, and high-performance building block for chronically implanted bioelectronics.
This means instead of having large implants encapsulated in thick metal boxes such as pacemakers, cochlear and brain implants, this technology could enable miniaturized, soft and fully biocompatible bioelectronics. They can also help us to create new bioelectronic devices that are not possible to make with existing technology due to various risk factors associated with chronic implantation of rigid and non biocompatible materials.
The team
We had to understand the challenges involved with diagnosis and treatment of patients with epilepsy, as well as the methodologies currently used. Therefore, this work required strong collaboration with our physician scientists at the Department of Neurology and the Institute for Genomic Medicine, in particular, Prof. Gelinas (co-corresponding author), the Director of the Epilepsy and Cognition Laboratory at Columbia University.
Also the work required careful microfabrication of complex transistors. Therefore, the clean-room facilities of Columbia Nano Initiative (CNI) was instrumental in this process.
Next steps
To make long-term implantable devices that can record and modulate the brain to help patients with neurological disease such as epilepsy.


































