Cancer is the leading cause of death, and the majority of cancer patients succumb to the disease due to the dissemination of malignant cells to other organs (metastases). While gene alterations (mutations) that cause inactivation of tumour suppressors or aberrant activation of tumour oncogenes have been involved in primary tumorigenesis, there is a dearth of mutations that are associated with cancer metastasis. MicroRNAs are short, ~22bp in length, RNA molecules that can post transcriptionally regulate the expression of mRNAs, mainly by suppressing their translation into proteins, and they have been shown to play an important role during the pathogenesis of various diseases, including cancer.
In a previous study, the group of Dr. Douglas Hanahan showed that miRNAs are differentially expressed during the distinct stages of tumour progression, such as proliferation, angiogenic switch, invasion, and metastasis, of pancreatic neuroendocrine tumours (PanNETs) (Olson, P., et al. Genes Dev 2009). In this study, we examined the role of a set of miRNAs that are upregulated in a subtype of PanNETs that is highly invasive and metastatic. Using a series of in-vivo assays, we found that miR-137 enhances tumour growth and local invasion, while the miR-23b cluster promotes metastasis to the liver.
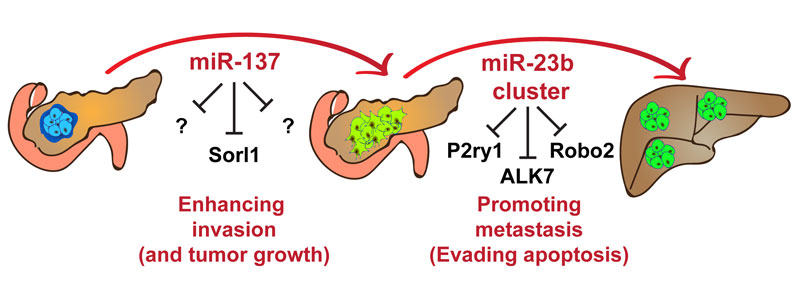
Figure: MicroRNA-137 enhances tumor growth and local invasion of pancreatic neuroendocrine tumors, while the microRNA-23b cluster promotes liver metastasis.
MicroRNAs have the ability to bind to several mRNAs, and therefore their gene targets are context-dependent. To find the gene targets of the miRNAs mentioned above, we developed a new algorithm, coined Bio-miRTa, which enables the identification of biologically-relevant miRNA gene targets. Using Bio-miRTa, we found that the miR-137 gene target Sorl1 has a tumour suppressor function, while we uncovered that the miR-23b cluster gene targets P2ry1 and Robo2 have an antimetastatic effect.
Moreover, our study revealed that the top miR-23b cluster candidate target is the Acvr1c/ALK7 gene. ALK7 is a member of the TGFß receptor superfamily, and we have recently demonstrated that the ALK7 signaling pathway has a tumour suppressor and antimetastatic activity by posing a pro-apoptotic barrier (Michael, IP., et al. Dev. Cell 2019). Using a series of in-vitro and in-vivo assays, we further characterized the regulation of ALK7 by the miR-23b cluster and demonstrated that the downregulation of ALK7 is necessary for the miR-23b cluster prometastatic activity. Finally, using Bio-miRTa in reverse, we identified a third family of miRNAs, miRNA-130/301, that might regulate the expression of activin B, the ligand of ALK7, during the initial stages of malignant transformation.
Overall, our study demonstrates the significance of miRNAs in the regulation of cancer progression and especially metastasis, and highlights the future therapeutic potential of targeting miRNAs.


































