Circadian clocks govern temporal coordination within and between organisms and are found across the tree of life, from cyanobacteria to humans. Clock oscillators are molecular feedback loops present in nearly every cell of the body and, via the rhythmic expression of genes every 24 hours, they drive rhythms in physiology and behaviour. This programmes an individual to do the correct thing at the correct time of day, ranging from cellular processes to tissue function to locomotor activity patterns. Failing to appropriately schedule activities may result in malnutrition, predation or missed mating opportunities.
When infected, hosts undergo a number of physiological changes – some caused by the pathogen, but some self-generated (e.g. lethargy, hypothermia, anorexia), – as an attempt to limit the damage caused by the infection. This includes disruption to host circadian rhythms in body temperature and activity patterns. Given that rhythms in physiological processes are usually tightly coordinated, why and how rhythms become decoupled requires explanation.
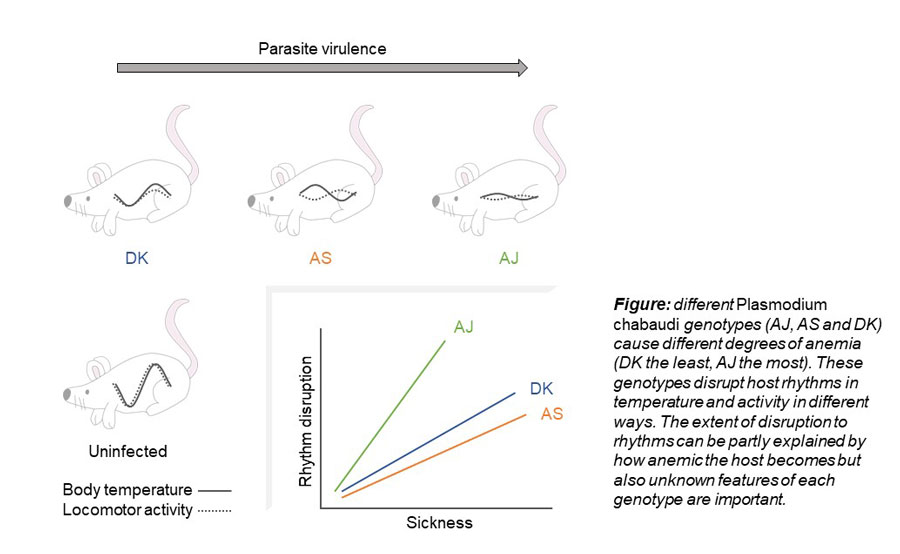
We tested the extent to which genetic variation amongst pathogens can explain variability in how host rhythms are affected during infection. We infected lab mice with one of three genotypes (genetic variants) of the same pathogen species (Plasmodium chabaudi, a rodent malaria parasite). These genotypes cause different severities of anaemia and other symptoms of malaria. We find that infection with each of the parasite genotypes changes host rhythms differently, for example the more virulent genotypes invert body temperature rhythms as mice approach peak sickness (see figure). Strikingly, we find that host sickness, measured as anaemia and parasite density in the blood, cannot entirely explain the amount of circadian rhythm disruption that hosts experience. This means that genetically variable features of parasites – yet to be discovered – influence host circadian rhythms.
We also find that body temperature and locomotor activity are disrupted independently. Such desynchrony between rhythms could cause a loss of metabolic regulation. Our study raises new questions about the costs and benefits of rhythm disruption for both host and pathogen, whether it is part of host sickness behaviour or if it enables pathogens to maximise their fitness.
Text by Kimberley Prior


































