Understanding how insects find and exploit floral resources is essential to maintain crucial ecosystem services of pollination. Insects like moths and butterflies unfurl their long and flexible modified mouthpart, the proboscis, and probe the flower to find the tiny nectary and feed from it, while hovering in front of it. Because the insect is largely airborne and hovering while feeding, this task requires exceptional motion control and precision as the insect must target a tiny nectary often the size of a few milimeters in the middle of the flower. Additionally, moths are generally active under low light conditions, in which their visual resolution is not sufficient to resolve the tiny nectary. Although most research has focused on how insects find their host plant, we know relatively little about the insect – flower interactions (Stöckl and Kelber, 2019). Recent research shows evidence that both visual and mechanical cues about the flower influence the success of moths in finding the nectary (Campos et al., 2015; Goyret, 2010; Goyret and Kelber, 2011) but how moths use their long, flexible proboscis to probe the flower and target the tiny nectary is unknown. Moreover, the insect proboscis is a unique, and novel sensory structure: it is actively controlled with muscles at the base and all along its length and it has tunable mechanics as it is fluid filled. Interestingly, the proboscis is richly embued with mechanical sensory neurons at the base and along its entire length.
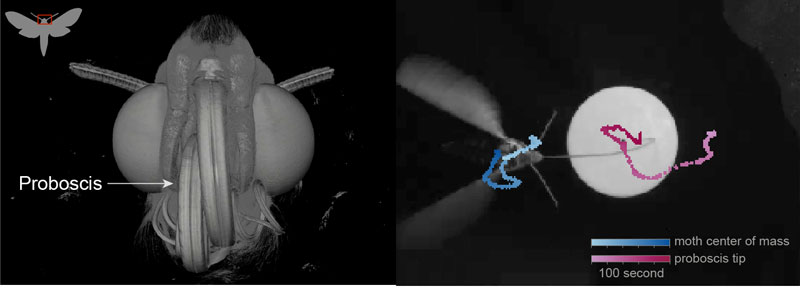
Figure: (Left) A microtomography reconstruction showing the frontal view of a Manduca sexta head with the proboscis coiled up. The inset shows the region (red box) of the moth which is imaged in the microtomography section. (Right) A still image from a video of a moth feeding from an artificial flower overlayed with the tracks of the proboscis tip (pink) and moth’s center of mass (blue).
The HFSP postdoctoral fellowship enabled us to capture, for the first time, the interaction of insect pollinators with 3D printed artificial flowers at high resolution. We built an automated behavioral rig that allowed flower naïve hawkmoths, Manduca sexta, to repeatedly visit a 3D printed artificial flower. 3D printing enables us to present novel floral shapes that differed only in one tactile feature: floral curvature, such that the flowers ranged from funnel shaped flowers, that were easy to exploit, to completely flat flowers that were more difficult. We also synchronized a video camera overlooking the flower from the top with tiny motion sensors in the flower nectary to measure the efficiency by which they emptied the flower. We found that the moths had the highest success rate with flowers that had funnel shaped curvature and the least success with the flat flowers. By measuring the how long it took moths to find the nectary over repeated visits, we found that hawkmoths got better at feeding from flowers within as few as three to five visits for all flowers, even those with slight curvatures that led to the nectary hole. However, they failed to learn to feed from flowers that were completely flat. Their ability to learn from flowers that provided mechanical cues to the nectary suggested that moths were not randomly probing the flower until they found the nectary but instead were using a strategy that allowed them to get to the nectary more efficiently.
We used machine learning-based pose extraction methods to track the proboscis tip as the moth was probing the flower to understand how the moths were using their proboscis to target the nectary. We found that moths systematically sweep their proboscis from flower edge-to-center-to-edge until they found the nectary. These active movements were highly reminiscent of rat whisking – where rats rhythmically sweep their whiskers on external surfaces such as walls to capture tactile information. The number of sweeps was generally higher for flowers that were difficult, like almost-flat or flat flowers, but as they learned to exploit these flowers, moths could find the nectary within a few sweeps. For the flat flower, however, they continued sweeping for several cycles. We also computed proboscis trajectory orientation with respect to the flower radial axis to understand how their strategy changes over repeated visits. We found that as moths learn, they specifically moved their proboscis along the radial axis i.e., along the curvature of the flower allowing them to target the nectary more efficiently. However, for flat flowers that did not have a slope the moths continued to probe everywhere and did not show evidence of a learnt strategy.
These results highlight the role of a novel structure, the proboscis, in tactile sensing and learning. In summary, the capacity for the moths to exploit tactile cues from flowers by actively moving their proboscis to extract these tactile features and learn efficient strategy to target the tiny nectary hole underlies their ability to feed from their host plant and hence provide crucial agro-ecosystem services of pollination.


































