The cortex is involved in many aspects of perception, cognition, and movement, and both receives sensory input and sends out motor commands. The cortex does not act in isolation however, instead sharing its information with an intermediate set of structures termed the basal ganglia. The basal ganglia receive connections from the entire cortex arranged along an orderly mapping. The basal ganglia in turn send information both back to the associated cortical regions and also to other brain structures, but whereas the cortex excites other regions, the basal ganglia inhibits other regions. These two systems, the excitatory cortex and the inhibitory basal ganglia, are poised to provide a push-pull influence across the brain. These opposing forces may be utilized for complementary purposes, with the cortex and basal ganglia carrying unique signals, or they may be cooperative, balancing each other at any given moment.
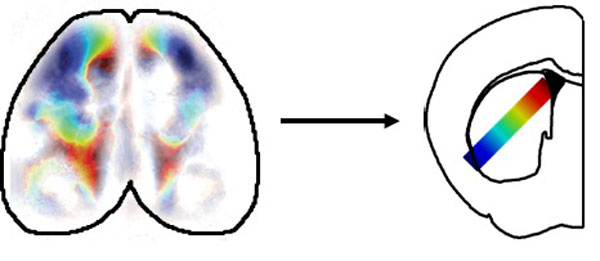
Figure: the cortex and striatum shared activity across a gradient of regions (colours).
We investigated the relationship between activity in the cortex and basal ganglia in mice by simultaneously recording wide regions of the cortex and the striatum, the input nucleus of the basal ganglia. This was achieved by combining wide-field calcium imaging, reporting bulk activity of large populations of neurons using fluorescence, with electrophysiology using Neuropixels probes, a new generation of high-density electrical recording arrays. During recording, mice performed a task in which a visual stimulus prompted them to move a wheel in a particular direction to receive a water reward.
We found that activity in the cortex and striatum was correlated in a spatially precise manner, with regions of the striatum being paired with specific regions of the cortex. This mapping followed previously described anatomical connections, indicating that activity effectively flows from the cortex to connected regions of the striatum.
Having established a mapping from the cortex to the striatum, we investigated whether the two structures exhibited similar or different activity patterns. During the task, activity related to stimuli, movements, and rewards was shared across connected cortical and striatal regions. Likewise, even when mice were passively resting, spontaneous activity was also shared between connected regions. The exception to this rule was a particular sparse cell type in the striatum, which is known to receive relatively weaker cortical input and likewise exhibited unique activity patterns.
The relationship between cortical and striatal activity was modified by learning, where the mapping between structures was consistent but untrained mice had much weaker stimulus responses specifically in the striatum. This suggests that activity does not always flow effectively from the cortex to the striatum, but instead only does so after it is made relevant through learning.
Together, these results indicate that the cortex and striatum act cooperatively by sharing activity patterns, and that learning enhances the transfer of activity from the cortex to the striatum. The cortex and basal ganglia therefore do not appear to serve separate roles, but instead provide continuously matched activity to downstream structures. This work contributes to a fundamental understanding of how the cortex and basal ganglia function together, which in the future can be applied towards basal ganglia related diseases like Parkinson’s, Huntington’s, addiction, and obsessive-compulsive disorder.


































