In early embryos, before cellularization, the cytoplasm is self-organized into distinct compartment-like regions. During this phase, embryos divide rapidly and with remarkable precision, requiring the cytoplasm to be partitioned in a highly orchestrated manner even in the absence of physical boundaries such as cell membranes. How such a robust organization emerges under these extreme conditions remains a fundamental question.
The self-organization of the cytoplasm is driven by microtubule asters, radial networks of filaments that fill the embryo and, through their interactions, define compartment boundaries. Although cytoplasmic partitioning was described more than a century ago, the mechanisms underlying compartment formation and stability have remained poorly understood. HFSP Cross-Disciplinary Fellowship awardee Melissa Rinaldin and her colleagues uncovered that cytoplasmic compartments are surprisingly unstable. By combining experiments in cytoplasmic extracts, living embryos, and modeling, the researchers showed that this instability leads to the invasion of neighboring compartments by microtubule asters and ultimately to compartment fusion. The authors demonstrate that the invasion process arises from the growth and interaction of microtubule asters. Asters grow through autocatalytic microtubule nucleation, in which new microtubules are nucleated from pre-existing ones. When combined with local inhibition at interfaces between neighboring asters, this growth mode can generate instabilities at compartment boundaries.
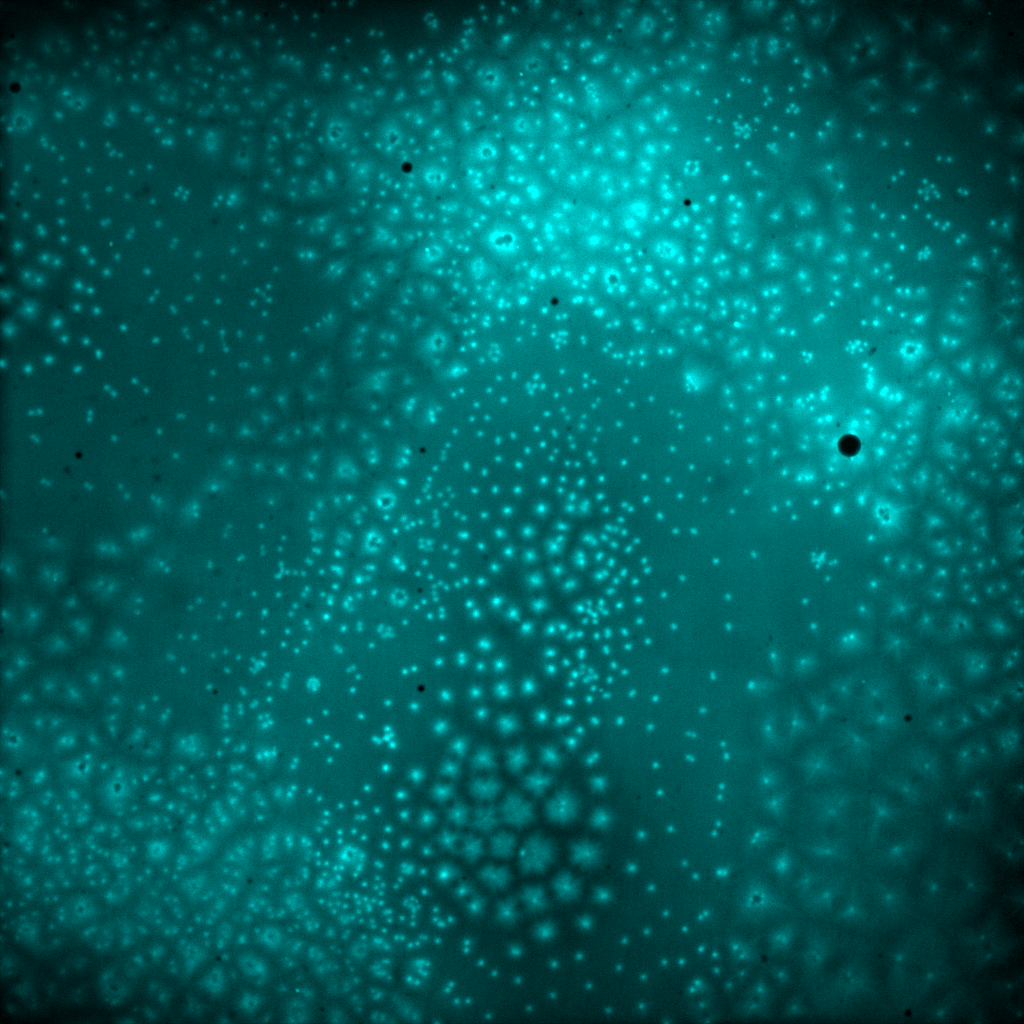
Remarkably, embryos have evolved strategies to suppress this instability. The team found that cytoplasmic organization can be stabilized either by matching the cell cycle duration to the instability timescale or by reducing the rate of microtubule nucleation. Comparing different species revealed distinct solutions: zebrafish, frog, and fruit flies have similarly sized embryos but markedly different aster architectures. For large asters, such as those observed in frog extracts and zebrafish embryos, the timing of cell divisions is precisely matched to the timescale of the instability. Divisions occur faster than the time required for the instability to unfold. In contrast, in fruit fly embryos, the rate of autocatalytic microtubule nucleation is lower, leading to smaller, stable asters that gradually fill the cytoplasm over multiple cell cycles. These findings suggest that regulation of microtubule nucleation may act as an evolutionary “dial” for controlling cytoplasmic patterning during early development.
By revealing how biological timing and physical instabilities are coordinated, this work highlights a fundamental principle underlying robust self-organization in living systems, with implications for embryonic development across species and for understanding how physical rules shape biological organization.


































