The cell cycle has been previously found to modulate actin filament assembly via metaphase Cdk1. However, several important questions about the function of Cdk1 in actin assembly regulation remain unanswered, such as what actin binding proteins are phosphorylated by the metaphase Cdk1, and how they are phosphoregulated.
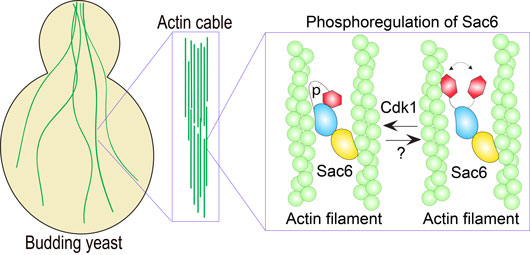
Figure: Phosphorylation of fimbrin (Sac6) by Cdk1
Interestingly, Cdk1 phosphorylation sites are usually mapped into the intrinsic disorder region (IDR). Many of the actin binding proteins are highly enriched in IDR, which is a flexible region for multiple purposes, such as regulation of protein-protein interaction and conformational change. In the reported work, Dr. Miao and his colleagues found that the cell cycle regulates actin binding protein fimbrin via phosphorylation on IDR by Cdk1, which causes the conformational change and regulates its interaction with actin filament in budding yeast.
The reported phosphoregulation study of actin crosslinking protein will inspire future work on studying the mechanisms of cell cycle regulation in actin filament assembly. In addition, this report also has a broader implication in the study of the physiological and pathological signal pathways that regulate actin filament assembly and the corresponding biological processes.
Reference
Fimbrin phosphorylation by metaphase Cdk1 regulates actin cable dynamics in budding yeast..Yansong Miao, Xuemei Han, Liangzhen Zheng, Ying Xie, Yuguang Mu, John R. Yates III & David G. Drubin. Nature Communications (2016) 7, Article number: 11265. doi:10.1038/ncomms11265.


































