Errors in chromosome segregation are the primary cause of infertility, miscarriage, and congenital aneuploidies, and their occurrence increases significantly with female reproductive age. In mammalian eggs, this age-related rise in aneuploidy has long been associated with the gradual decline of chromosome cohesion, the molecular glue that keeps sister chromatids together during meiosis. However, because cohesion deteriorates slowly over decades in humans, it has been challenging to distinguish cause from effect or to understand why aneuploidy rates rise rapidly late in reproductive life.
In this study, HFSP Research Grant Awardee Binyam Mogessie and her team created a new experimental system that enables rapid and precise reduction of chromosome cohesion in otherwise young mouse eggs. By combining CRISPR-based genome editing with targeted protein degradation technologies, they developed a mouse model in which the meiosis-specific cohesin subunit REC8 can be quickly removed on demand. In the context of meiosis, this method compresses decades of reproductive aging-related chromosomal deterioration into hours, resulting in aging-like chromosome segregation defects without using aged animals. The system allows direct observation of cohesion dynamics in live eggs and provides precise control over the timing and severity of chromosome segregation errors.
Using this platform, the scientists demonstrate that partial loss of chromosome cohesion is enough to cause premature separation of sister chromatids, a key chromosomal defect found in aged eggs. Notably, the ability to quickly and precisely lower cohesion provides the first direct experimental proof of a critical threshold beyond which chromatid separation becomes excessive. In animals that age naturally, where cohesion decreases gradually over years or decades, it has remained unclear where naturally aged eggs stand relative to this threshold, or whether the sharp increase in aneuploidy occurs after crossing it. This system now enables direct investigation of that relationship and allows individual age-related factors to be examined separately and in specific combinations. As a proof of principle, the team showed that modest depletion of the centromeric histone variant CENP-A, at levels similar to those seen in naturally aged eggs, exacerbates chromosomal instability when cohesion is compromised, indicating that age-related aneuploidy can result from the combined weakening of multiple chromosomal maintenance pathways, not just cohesion loss.
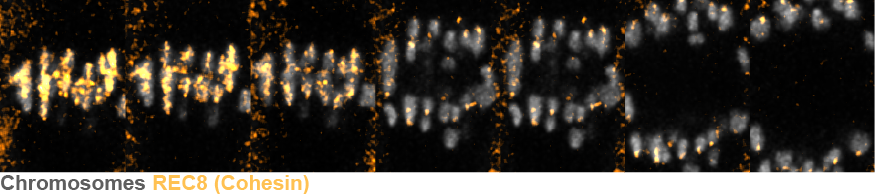
Beyond the specific biology revealed here, this work presents a widely applicable framework for studying egg aging and infertility. By eliminating the need for long-term animal aging studies, the system reduces experimental barriers, decreases animal use, and enables systematic, mechanistic investigation of chromosome maintenance pathways in the female germline. It also paves the way for unbiased screening methods, including small-molecule or targeted degradation libraries, to identify factors that maintain chromosome integrity in eggs.
As maternal age at first pregnancy continues to increase worldwide, understanding how and why eggs lose chromosomal organization has become more important for reproductive health. This work offers both a conceptual advance and practical tools for identifying the cellular causes of age-related aneuploidy, with the long-term aim of developing strategies to enhance egg quality and reproductive success.


































